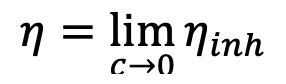
My question is - as I have my viscosity vs concentration graph a straight line, how will dividing each viscosities again by their respective concentration give another straight line as the quotient is different in this case? Or did I calculate the reduced viscosity incorrectly ( the formula I provided above). My professor asked me to prepare another solution as according to him this is not normal and that reduced viscosity should also vary more or less directly with the concentration. When you squeeze a non-Newtonian slime, like oobleck, or pound it with your. What this means is, when you pour slime or let it ooze through your fingers, it has a low viscosity and flows like a thick liquid. Where $\eta_0$ is the average viscosity of the solution, $\eta_s$ the average viscosity of the solvent and $c$ the concentration of the solution. The technical explanation is that slime is a fluid that changes its ability to resist deformation according to shear or tensile stress. To calculate the reduced viscosity, I used following relation : It means that a molecule of a liquid should gain sufficient energy to push aside the surrounding molecules before it can take part in the liquid flow. 
Viscosity measurements are made in conjunction with product quality and efficiency. As we all know that viscosity is nothing but the measure of a substance’s resistance to the motion under an. The unit is usually expressed in poise or centipoise. Brookfield deals with liquids and semi-solids. The coefficient of viscosity is defined as the force of friction that is required to maintain a difference of velocity of 1cm/s between parallel layers of fluid. Viscosity is a principal parameter when any flow measurements of fluids, such as liquids, semi-solids, gases and even solids are made. Calculation: Calculate the viscosity by the relationships: 1 2. Viscosity (symbol: ) is a measure of a fluid’s internal resistance to flow. The force necessary to move a plane of area A past another in a fluid is given by Equation 2.6.1 2.6. 
In short, the values for reduced viscosities are not directly proportional to the concentration as was in the case of viscosity. Find Viscosity-related information at AMETEK Brookfield Customer Learning Center. However, when I plot a graph of reduced viscosity vs concentration, in this case I find that the points are not in a line but are scattered. When I plot concentration vs viscosity graph in the excel, I have a perfect line that is the average viscosity is directly proportional to the concentration. I prepared 3 different polymer solutions with varying concentration of 100, 50 and 25 ppm in order to measure their viscosity. Reynold proved that the critical velocity for a liquid flowing in a tube is vc k/a, where is density and is viscosity of the liquid, a is radius of the.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
